Liver diseases are a significant cause of death:
- They are responsible for more than 50,000 deaths annually in the United States.
- They cause more than one million hospitalizations each year.
- They cause more than 100,000 cases of end-stage liver failure annually.
Orthotopic liver transplantation (OLT) is a limited solution:
- It is the only curative treatment currently available for patients with irreversible liver failure.
- It is limited by a small number of donors.
- It involves significant risks, such as infection, rejection, and death.
- It is very expensive.
Hepatocyte transplantation may be an alternative solution:
- This approach may provide effective treatment for patients who are not suitable for OLT.
- It may improve control of portal hypertension.
- It may increase functional liver mass.
Lymph nodes as an alternative site for hepatocyte transplantation:
- Previous research showed successful hepatocyte transplantation in mouse lymph nodes.
- A groundbreaking study examined the feasibility of this approach in a pig model of liver failure.
Research methods:
- 12 pigs underwent induction of subacute liver failure using a low-protein diet and toxic hepatitis.
- Autologous hepatocytes were isolated from the left lobe of the liver using a special process called "reverse flow surgery."
- Hepatocytes were injected directly into the mesenteric LNs using a special needle.
- 6 pigs served as a control group and received a sham injection.
- The animals were monitored for 60 days.
Research results:
- Hepatocyte transplantation into LNs was safe and feasible.
- Liver cells successfully engrafted in the LNs within 7 days.
- Functional ectopic liver tissue formed within the LNs.
- The amount of ectopic liver tissue was proportional to the severity of damage to the original liver.
- A significant decrease in portal blood pressure was observed.
- Liver function significantly improved in the treatment group compared to the control group.
- No serious side effects were observed.
Research conclusions:
- Hepatocyte transplantation into LNs is a new and promising therapeutic approach for treating liver failure.
- This approach may provide an effective and more accessible solution for patients with end-stage liver disease.
Further research:
- Future studies will focus on:
- Developing more efficient transplantation techniques.
- Developing drugs and support for ectopic liver tissue.
- Examining the efficacy and safety of this approach in humans.
LyGenesis: An Innovative Solution for Mini-Livers, Human Trials.
LyGenesis is a biotechnology company developing a groundbreaking technology for growing "mini-livers" in the lymph nodes of patients suffering from end-stage liver failure. This approach may provide an innovative solution to the problem of organ shortage for transplantation and save many lives.
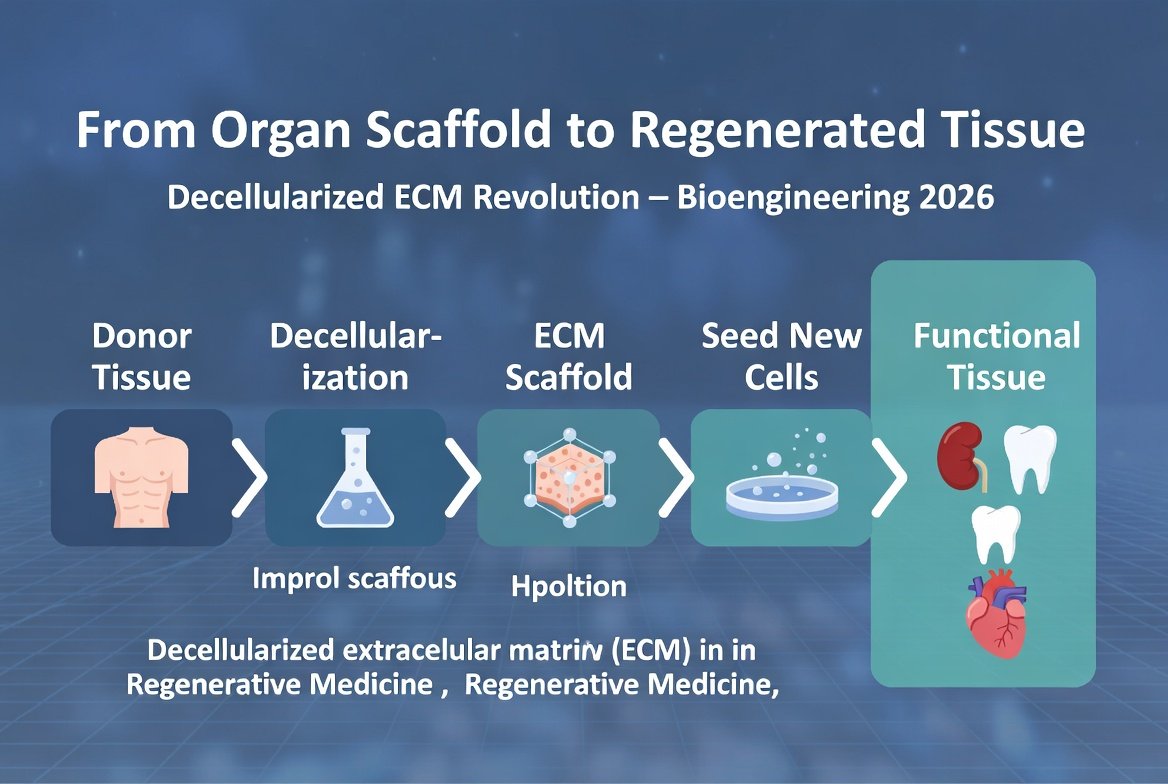
LyGenesis technology:
- The company uses donated livers that are not suitable for standard transplantation.
- Liver cells are separated from the liver and processed in the lab.
- Liver cells are injected through a tube in the throat into the patient's lymph nodes.
- The lymph nodes provide a supportive environment for the growth of liver cells.
- Within a few months, functional "mini-livers" form inside the lymph nodes.
Advantages of the LyGenesis approach:
- A solution to the problem of organ shortage for transplantation.
- More accessible treatment for patients suffering from liver failure.
- A relatively simple procedure compared to standard liver transplantation.
- Relatively low risk of rejection and infection.
- Potential to improve the quality of life and life expectancy of patients.
Clinical trials:
After a successful Phase 1 clinical trial in humans,
LyGenesis received FDA approval to begin a Phase 2 clinical trial in humans.
The trial will examine the safety and efficacy of the treatment in 12 patients.
Preliminary results are expected to be published in 2026.
It should be noted that LyGenesis plans to perform similar procedures for a variety of different organs, here is their progress: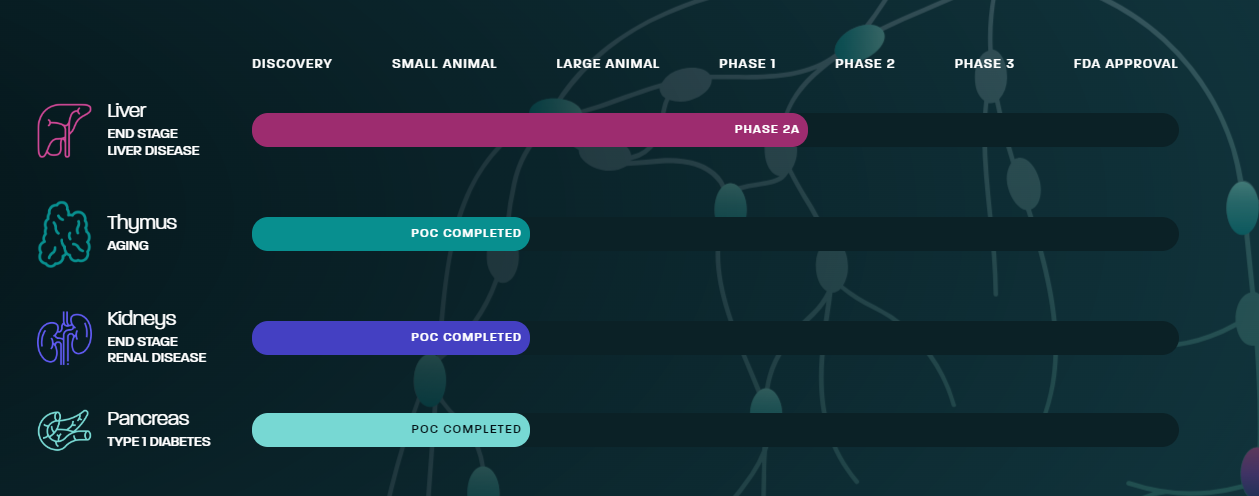
LyGenesis's technology for growing "mini-livers" in lymph nodes is an innovative and exciting solution to the problem of organ shortage for transplantation.
This approach may save many lives and improve the quality of life for patients suffering from end-stage liver failure.
.
References:
https://www.lygenesis.com/
https://journals.lww.com/lt/fulltext/2020/12000/development_of_ectopic_livers_by_hepatocyte.14.aspx
https://pubmed.ncbi.nlm.nih.gov/23000933/

💬 תגובות (0)
היו הראשונים להגיב על המאמר.