If you ever wondered what the "next step" of anti-aging looks like, the answer came this week from Nature. The world's leading scientific journal reported that the most powerful method we have ever identified for reversing cellular aging - partial reprogramming - is about to be tested in humans for the first time. This is not speculation. It is happening.
The Story of Yamanaka Factors
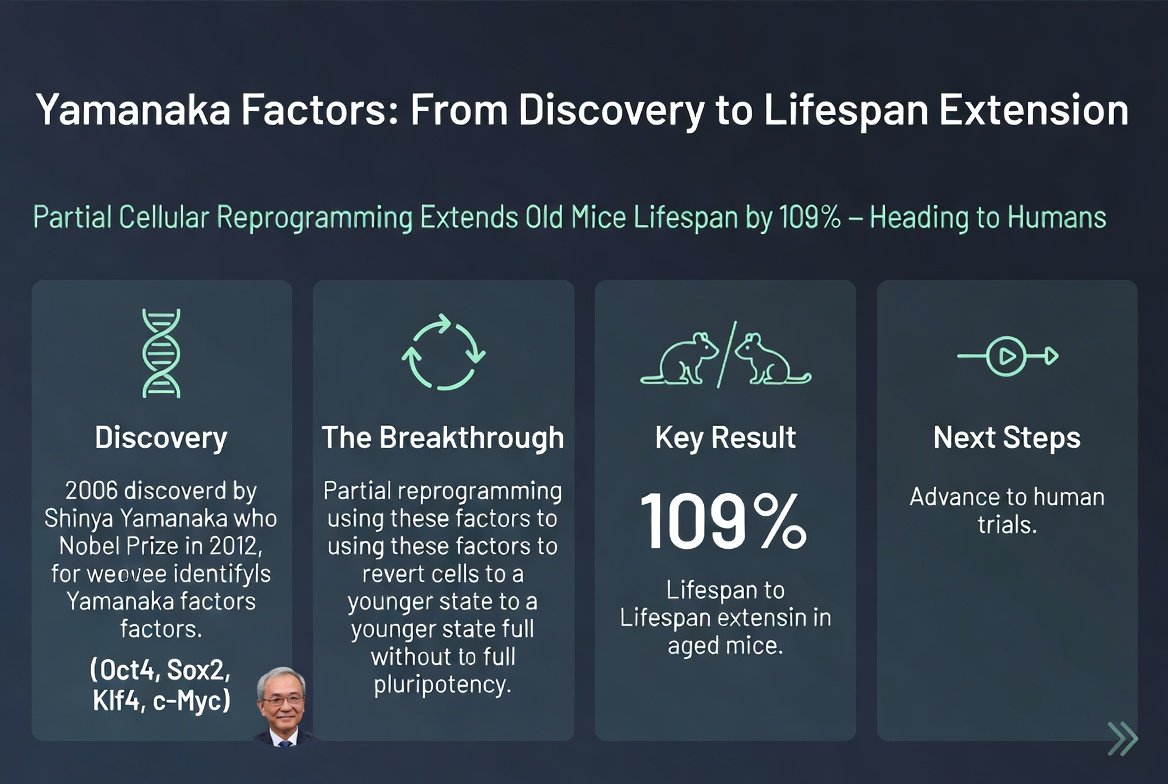
In 2006, a Japanese researcher named Shinya Yamanaka attempted a task considered impossible: reverting an adult cell to a stem cell state. He searched for the genes that make cells stem-like, and over time narrowed the list to just 4 genes: OCT4, SOX2, KLF4, MYC. When he introduced these four genes into an adult cell - they reverted it backward, creating induced pluripotent stem cells (iPSCs).
The discovery earned him the Nobel Prize in 2012. But there was a problem: the cell returns to a stem cell fully. If you activate the genes in a skin cell, the cell will revert to a stem cell - not a young skin cell. This is not an anti-aging process - it is an "erasure" process.
The Shortcut: Partial Reprogramming
In 2016, another researcher named Juan Carlos Izpisua Belmonte from the Salk Institute made a critical change: he activated the Yamanaka factors for only a short time. Instead of days - hours. Then he stopped.
The result was astonishing: the cells did not revert to stem cells. They remained skin cells, liver cells, or muscle cells. But - they aged backward. Their epigenetic clock rewound. Aging markers disappeared. Their function improved.
Izpisua demonstrated this in several mouse experiments, and then in 2020 showed that partial reprogramming made old mice physically younger. They performed better in memory tests, restored regeneration in damaged tissues, and improved vision quality.
Altos Labs: $3 Billion to Develop a Human Treatment
In 2022, a company called Altos Labs was founded, funded by Jeff Bezos, Yuri Milner, and other billionaires - $3 billion in initial funding. The goal: to move partial reprogramming from the lab to the human clinic. Its research team includes Izpisua himself, as well as Shinya Yamanaka, and over 200 top-tier researchers.
For 4 years, the team worked behind the scenes. Now, in 2026, Nature reports: they are ready for humans.
The First Clinical Trial: What Does It Focus On?
The first trial will not be a general "anti-aging drug." To get FDA approval for such a novel technology in humans, a specific indication with urgent medical need must be chosen. The researchers chose vision restoration in glaucoma patients:
- How: Direct injection into the eye of an AAV vector (a delivery virus) carrying the Yamanaka factors (without MYC - it is a cancer risk).
- Why glaucoma: A disease causing gradual death of retinal ganglion cells. Currently, no treatment restores dead cells.
- How controlled: Gene expression occurs only when the patient takes a drug (doxycycline) - allowing the process to be turned off at any moment.
- Trial size: 30 patients, ages 50-80, with advanced glaucoma.
- Timeline: 24-month follow-up after the first injection.
What Could Be Learned from Success (or Failure)
If the trial succeeds, it will prove 3 revolutionary things:
- Humans can tolerate partial reprogramming without developing cancer - the main risk.
- Old cells in humans are capable of rejuvenation - not just in mice.
- The approach is scalable - heart, liver, brain, skin - all tissues could be candidates for a similar approach.
If the trial fails, we will learn the limits of the approach - perhaps variations of factors or more controlled delivery methods will be needed.
Remaining Dangers
The team does not hide the concerns:
- Cancer: If cells are reverted too far "backward," they could become stem cells - and stem cells in the eye have the potential for teratoma (a tumor containing different tissues).
- Loss of cell identity: Retinal ganglion cells that undergo high-intensity reprogramming could lose their neural connections, thus impairing vision instead of improving it.
- Immune response: The AAV viral vector could trigger a local immune response.
The Broader Perspective
If we think about Aubrey de Grey's 7 damage theory we wrote about two weeks ago, partial reprogramming is a direct response to several of them simultaneously - it resets not only epigenetic damage but also mitochondrial function and improves cellular renewal. This is why the scientific community sees it as perhaps the most powerful of all anti-aging approaches identified so far.
If the eye trial succeeds, the next steps are likely within 5-7 years: trials in the heart (after a heart attack), muscle (sarcopenia), and brain (Parkinson's, Alzheimer's). If everything goes well - in 15-20 years we may see partial reprogramming treatments as the standard of care for elderly patients. And after that - who knows? Perhaps also for those who are not sick.
What This Means for You Now
Nothing directly. If you are 50+, the drug will not be available before you are 65. If you are 30, there is a high likelihood you will see revolutionary treatments in your later years. The best thing you can do now is to maintain your body until the treatments arrive: nutrition, physical activity, sleep, and most importantly - avoiding damage that will be hard to repair (smoking, harmful sun exposure to skin, chronic stress).
We are living in a special moment in the history of the human species. This is not hyperbole.
References:
Nature - Cellular Reprogramming in Humans
Altos Labs

💬 תגובות (0)
היו הראשונים להגיב על המאמר.