The tooth is an incredibly complex organ: hard tissue (enamel and dentin), living tissue (dental pulp with nerves and blood vessels), a periodontal ligament, and complete dependence on very precise development in childhood. When such an organ is lost, the solution of modern dentistry has been dentures and crowns. But what if we could simply grow a new one from stem cells? A new review published this week in the scientific journal Cureus systematically surveys all the approaches advancing toward this goal.
Why Tooth Regeneration is the Holy Grail
The standard implant—a titanium screw placed in the jaw with a porcelain crown—works well, but it has limitations:
- No living tissue: The implant does not feel pressure or heat, and does not connect to a nerve.
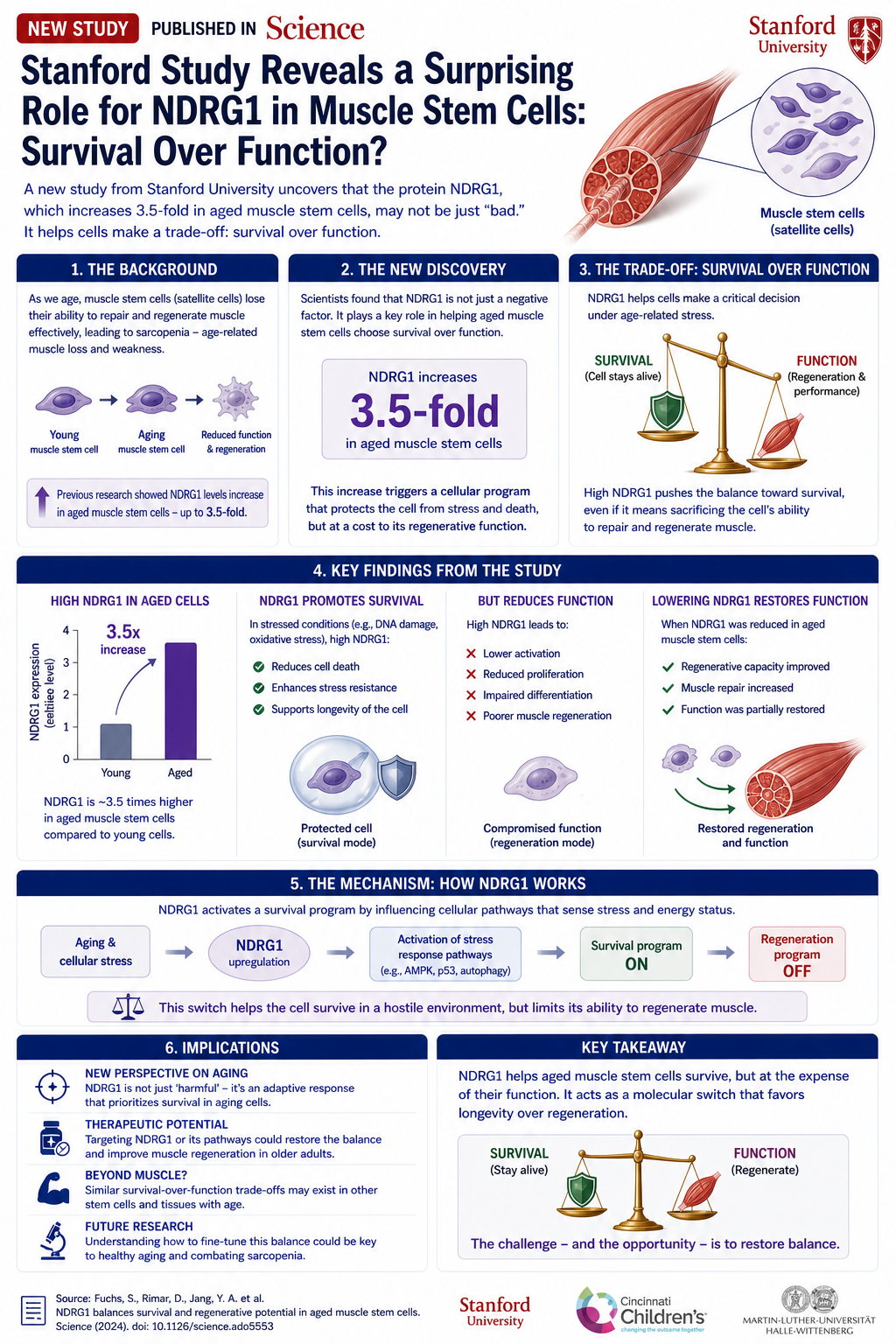
- Bone loss: Without a living tooth root, the surrounding jawbone begins to resorb.
- Infection risk: Peri-implantitis is a common problem in the first decade.
- Limited lifespan: An implant typically lasts 15-25 years. A biological tooth—for life.
A new tooth that grows biologically would solve all these problems. The question is how.
The Five Types of Dental Stem Cells
The review distinguishes between five types of stem cells, each of which can contribute to a different part of the tooth:
- DPSCs (Dental Pulp Stem Cells): Isolated from the dental pulp of adults. Multipotent: can become odontoblasts (the cells that produce dentin), neurons, or endothelial cells. The gold standard in research.
- SHED (Stem cells from Human Exfoliated Deciduous teeth): Stem cells from shed "baby teeth." Younger and with higher proliferation potential than DPSCs.
- SCAP (Stem Cells from Apical Papilla): From the developing root tip. Capable of producing thick primary dentin.
- PDLSCs (Periodontal Ligament Stem Cells): From the ligament that holds the tooth. Essential for anchoring the new tooth to the bone.
- DFPCs (Dental Follicle Progenitor Cells): From the follicle surrounding the developing tooth. Can produce cementum (the material covering the root).
The Biological Scaffold
Stem cells alone will not form a tooth shape. They need a 3D scaffold to guide where to grow and in which direction to differentiate. The review covers three families of scaffolds:
- Synthetic polymers: PLA, PLGA, PCL. Can be precisely shaped in 3D, degrade at a known rate. The downside: not always cell-friendly.
- Natural scaffolds: Collagen, chitosan, hyaluronic acid. Cell-friendly but difficult to shape precisely.
- Decellularized matrices: A tooth from an external source with all cells removed, leaving only the protein structure. The newest step—the scaffold "remembers" the original tooth shape.
Growth Factors That Activate the Process
Cells on a scaffold still do not form a tooth. Chemical signals are needed to instruct them to divide, differentiate, and organize themselves:
- BMPs (Bone Morphogenetic Proteins): Especially BMP-2 and BMP-4. Activate the mineralization process.
- Wnt signaling: The same pathway that worked in the Chinese SMAD7 study. Controls tooth position and shape.
- FGF (Fibroblast Growth Factors): Promote proliferation and blood vessel formation.
- TGF-β: Controls dentin formation and epithelial-mesenchymal interaction.
What Works in the Lab Today
The review documents several impressive preclinical successes:
- Japanese researchers succeeded in growing a complete tooth with root, pulp, enamel, and dentin in mice, using a combination of DPSCs with embryonic epithelial cells.
- An American study showed regrowth of damaged dental pulp in dogs using SCAP injection.
- A Chinese group demonstrated periodontal ligament growth from PDLSCs—the critical step for anchoring.
Challenges Delaying the Clinic
Why is this still not at your dentist?
- Vascularization: A tooth needs a blood supply through microscopic channels at the root tip. Creating a functional blood vessel network inside a scaffold is the hardest step.
- Innervation: How to get the trigeminal nerve to "connect" to the new pulp? Still unsolved.
- Integration with jawbone: The tooth must be held in the bone with the right strength. Too fast—difficulty. Too slow—collapse.
- Time: A tooth takes 6-12 months to develop in children. Will patients wait?
- Cost and mass production: How to turn a complex lab process into something available to patients.
What About 5 More Years?
The review's conclusion is cautious but optimistic. The technology to grow a biological tooth in the lab already exists. The gap is engineering and clinical, not theoretical. Leading teams in Japan, China, and the USA expect Phase-1 human trials within 5-7 years. Until then, implants are still with us—but for the first time, they are not the only good solution on the horizon.
References:
Cureus Journal of Medical Science

💬 תגובות (0)
היו הראשונים להגיב על המאמר.