If aging and cancer were dancing a duet, telomerase would be the orchestra. This enzyme is responsible for rebuilding the telomeres at the ends of chromosomes, and without it, stem cells would age and the growth potential of cells would end. The problem: In about 90% of cancer types, telomerase is forcibly activated and allows cancer cells to divide indefinitely. An international team, which published its findings in Science in March 2026, presents for the first time a complete 3D map of the enzyme, and within it a surprising discovery: a surface structure we didn't know about, which could be a target for a new generation of cancer drugs.
Why is Telomerase So Important?
Telomeres, repetitive DNA sequences at the ends of chromosomes, shorten with each cell division. When they erode enough, the cell loses its ability to divide (senescence) or dies (apoptosis). This is a natural process that protects us from cancer: a cell that has grown too much meets its fate.
But there is a flaw in this defense. In 90% of cancer types, the TERT gene (which produces telomerase) is reactivated. Cancer cells can lengthen their telomeres without limit, becoming immortal. This is one of the "six hallmarks of cancer" identified in the early 2000s.
The Problem: Hiding the Complete Picture
For decades, scientists documented telomerase in parts: only the protein component, only the RNA, only part of the complex. The reason: the enzyme is complex, its parts are soft, and in an electron microscope it moves and disperses. You can't develop a targeted drug if you don't see the full shape.
Since early 2025, only two teams managed to see parts, but no team succeeded in revealing the complex: TERT (the protein), TER (the RNA), Est3 (the helper protein), all together.
The Breakthrough: International Collaboration
The team, led by researchers from the University of Montreal, UCLA, and others, used Cryo-EM (cryogenic electron microscopy). They froze the enzyme in ultra-thin ice, photographed it from millions of different angles, and calculated the complete shape at near-atomic resolution.
To simplify the experiment, they chose to work with telomerase from yeast (Saccharomyces cerevisiae) instead of human. Yeast is less complex, its telomerase structure is essentially similar to the human one, and it is easier to produce in the lab. This was the step that enabled the revolution.
The Discovery: A Secret Zinc Finger
When the structure was revealed, the team identified something no one had described before: a zinc finger within telomerase. Zinc fingers are structural motifs in proteins that precisely grasp DNA or RNA. Until now, we didn't know telomerase used one.
The more preliminary discovery: this finger doesn't just grasp RNA, it activates the enzyme. Without it, telomerase exists but doesn't work. With zinc in place, it takes off into action.
"This is a puzzle piece no one knew was missing. Now it's clear how telomerase is activated at the right moment, and how it is turned off when needed."
Est3: The Scaffold Holding It All Together
The team also discovered the true role of Est3, a protein everyone knew about but didn't understand its function. In the new picture, Est3 is a molecular scaffold that connects all the components of telomerase and maintains its solid structure. Without it, the enzyme falls apart.
This is also a promising drug target: if you can dismantle Est3, you can eliminate telomerase entirely, without harming other proteins in the cell.
Why Is This Important for Cancer?
With this knowledge, pharmaceutical companies can develop drugs that do one of two things:
- Block the zinc finger: Reduces telomerase activation in a cautious manner. In cancer cells that rely on telomerase, this is disastrous. In healthy cells, the effect is minimal because they use telomerase to a tiny extent.
- Dismantle Est3: The drug that destabilizes the telomerase structure.
The first experiments in mice are planned for 2027. If all goes as planned, clinical trials in humans could begin in 2029-2030.
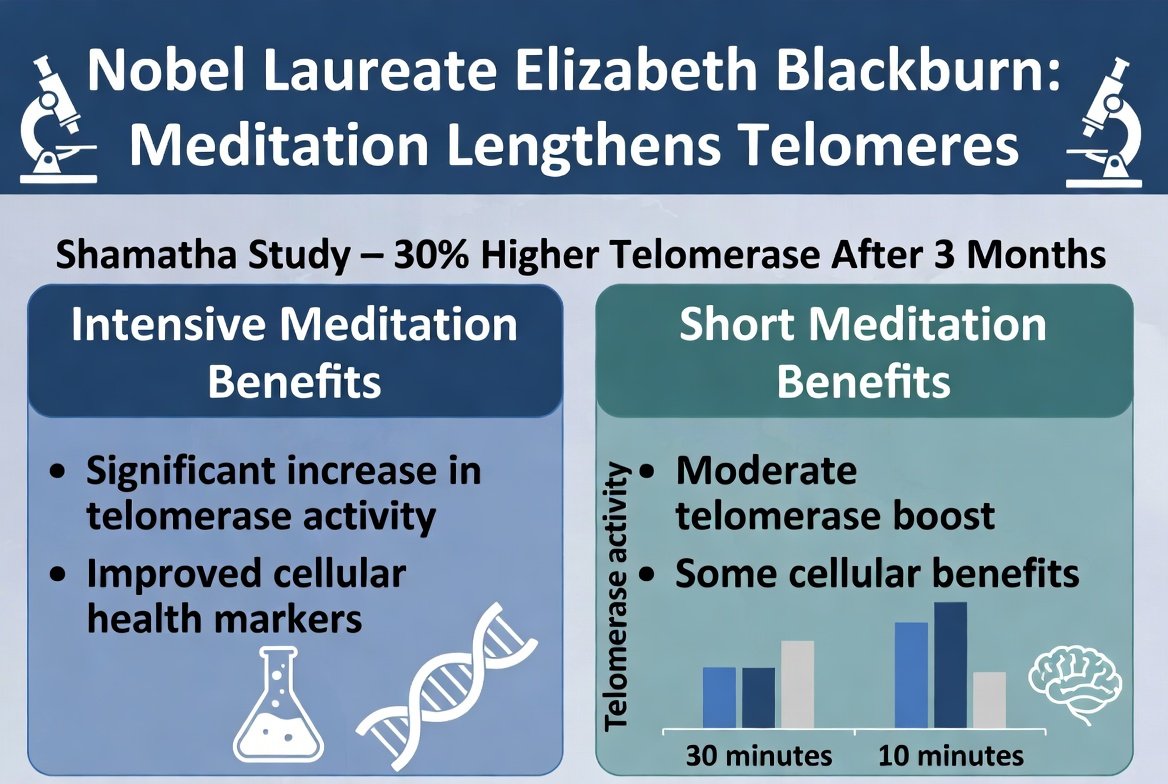
Implications for Anti-Aging
The other side of the coin: aging. Drugs that suppress telomerase help against cancer but can accelerate aging (less cell renewal). Drugs that activate telomerase can slow aging but risk cancer.
The new discovery opens the possibility for tissue-specific activation. A drug that activates telomerase only in specific stem cells (e.g., in the skin or blood), without reaching other cells, could provide the benefits without the risk.
Broader Context
This is an example of what scientists in the field call structure-based drug design. Instead of searching for drugs randomly, you look at the drug target in 3D and design a molecule that fits precisely. Most new drugs since 2010 have been developed this way. Now, finally, we have the tool to design drugs against telomerase.
This discovery unlocks decades of drug research. Until now, pharmaceutical companies tried to develop telomerase inhibitors blindly, and many failed. Now they have a map.

💬 תגובות (0)
היו הראשונים להגיב על המאמר.